
Nanocosmetic Regulation in 2026: EU, FDA, and ISO Standards for Sub-Micron Delivery Systems
Regulatory framework for sub-micron cosmetic delivery in 2026. EU Cosmetics Regulation 1223/2009 nano notification, FDA voluntary nano guidance, and ISO 29701 and 80004 standards governing NanoBase™ and tri-domain nano-delivery platforms engineered by Pensive Beauty Labs.
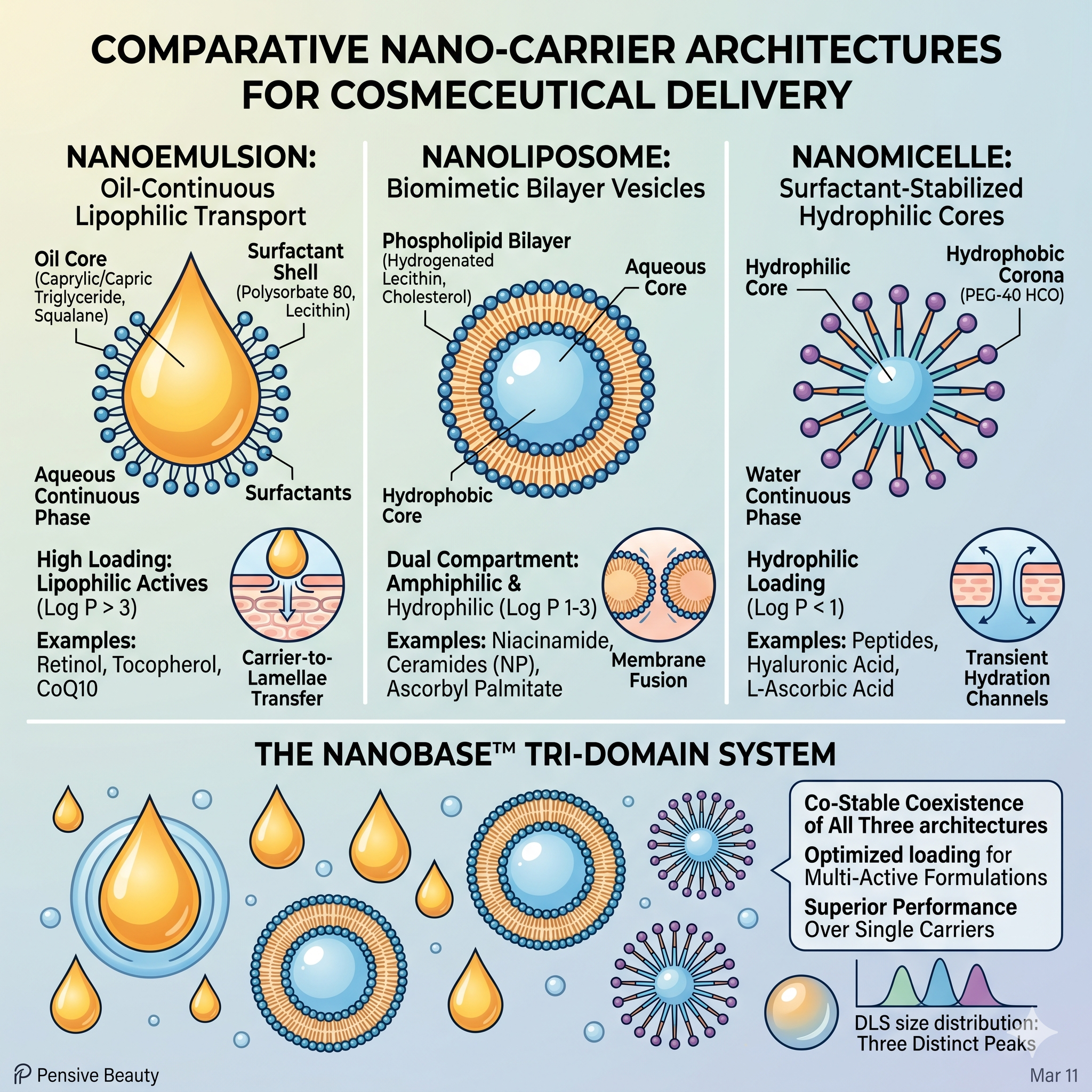
Nanoemulsion vs Nanoliposome vs Nanomicelle: Comparing Carrier Architectures for Cosmeceutical Delivery
Head-to-head technical comparison of three dominant nano-carrier architectures for cosmeceutical delivery: nanoemulsions (40 to 200 nm droplets), nanoliposomes (phospholipid bilayer vesicles), and nanomicelles (5 to 100 nm surfactant aggregates). Loading capacity, stability, stratum corneum penetration pathways, and why NanoBase™ tri-domain architecture from Pensive Beauty Labs outperforms single-class carriers.

How to Start a Skincare Line: A Complete Guide for 2026
Learn how to start a skincare line in 2026 with NanoBase™ nano-delivery technology. Step-by-step guide covering formulation, branding, regulatory compliance, and manufacturing partnerships.

The Complete Guide to Private Label Skincare Manufacturing
Complete guide to private label skincare powered by NanoBase™ nano-delivery technology. Learn how to launch your own branded product line with advanced tri-domain formulation architecture.

Custom Skincare Formulation Services: Build Your Brand with Nano-Precision Technology
Explore custom skincare formulation services built on NanoBase™ tri-domain nano-delivery architecture. From concept to finished product, advanced molecular engineering for next-generation topical delivery.

Nanotechnology in Skincare: The Future of Precision Beauty
How nanotechnology is transforming skincare delivery. Deep dive into NanoBase™ tri-domain architecture, nano-emulsion stability, and why traditional HLB emulsion systems fail at molecular-scale penetration.

The Complete Guide to Peptide Skincare Formulation: Overcoming Delivery Barriers
Complete guide to peptide skincare formulation and nano-delivery. Covers signal peptides, copper peptides, matrikines, and how NanoBase™ architecture optimizes peptide bioavailability through tri-domain encapsulation.

Why Most Skincare Actives Never Reach the Dermis
Conventional emulsions produce 1–100 µm carriers against a stratum corneum barrier with 10–50 nm intercellular spacing — a dimensional mismatch that precludes transepidermal transport. NanoBase™ tri-domain nano-delivery (125–195 nm, PDI ≤0.20) resolves all three HLB barriers simultaneously.

